NiaVasc™ (120 Tablets)
Sustained-Release Niacin (vitamin B3)
» Supports the Maintenance of Healthy Blood Lipids*
» Supports Normal Carotid Intima-Media Thickness (CIMT)*
$43.99
Description
Niacin is one of the most studied and documented nutrients for support of lipid levels already within the normal range, especially high-density lipoprotein cholesterol (HDL-C) levels. Sustained release niacin, as found in NiaVasc™, has a lesser flushing effect compared with instant-release niacin. Use of a proprietary, wax-coated technology permits release that is complete in seven to eight hours, the time that is considered ideal for a time release form of niacin.*
The cardiovascular benefits of niacin (vitamin B3) were introduced in the June 1956 issue of Mayo Clinic Proceedings. More than 20 years later, the Framingham Heart Study touted the benefits of niacin on lipid metabolism. A decade later, as the study continued, researchers labeled niacin “front-line” cardiovascular support. This status was further reinforced in 1988 when the National Cholesterol Education Program (NCEP) panel designated niacin a “first-line therapy” for support of specific parameters related to cardiovascular health.*
Why Isn’t Niacin More Widely Used? The two common concerns are cutaneous flushing and increased liver enzymes. Cutaneous flushing is harmless, although it can be a nuisance. Flushing is most often seen with the use of immediate/instant-release forms of niacin and can
occur with doses as low as 30 mg/day, but it is more likely to occur with the much higher doses used to support healthy blood lipids. Flushing may last 10 to 15 minutes and rarely, but possibly, up to two hours. The proprietary wax-coated technology used in NiaVasc tablets allows a gradual, sustained release of niacin over a seven-to-eight hour period. This delivery dramatically reduces the flushing associated with immediate-release forms. Adherence to a regimen with the special wax-coated form of niacin, as found in NiaVasc, ranged from 88-97% in four human clinical trials. Flushing, itching, tingling, and upper gastrointestinal side effects were minimal, but did increase when dosing was increased to 2000 mg/day. It is important to note that NiaVasc should not be confused with “no-flush” niacin, which is inositol hexanicotinate (IHN), a supplement that does not contain any free niacin and may not be as supportive of cardiovascular health as those providing nicotinic acid.*
The second concern with regard to liver enzyme elevation was first elucidated by the results of McKenney’s study, published in 1994 in
the Journal of the American Medical Association (JAMA), wherein subjects received 3000 mg/day of niacin over an extended period
of time. In April 2004, McKenney retracted his earlier warnings about the harmful effects of niacin and publicly supported its unique benefits. Although they generally do not enter an unhealthy range, liver enzymes may increase when initiating niacin therapy, especially in amounts greater than 1000 mg/day. Enzyme levels return to normal promptly after cessation of niacin.*
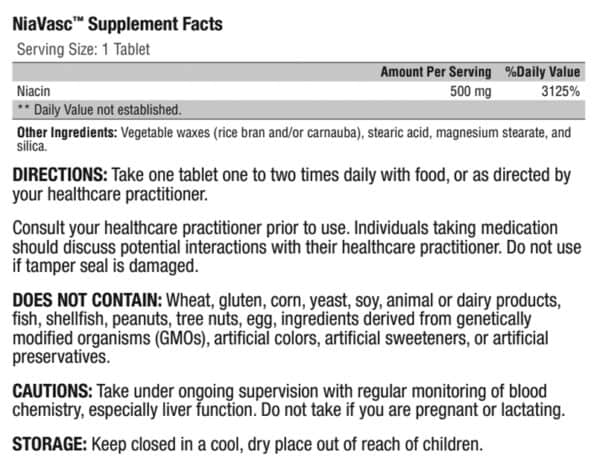
DIRECTIONS: Take one tablet one to two times daily with food, or as directed by your healthcare practitioner.
Consult your healthcare practitioner prior to use. Individuals taking medication should discuss potential interactions with their healthcare practitioner. Do not use if tamper seal is damaged.
DOES NOT CONTAIN: Wheat, gluten, corn, yeast, soy, animal or dairy products, fish, shellfish, peanuts, tree nuts, egg, ingredients derived from genetically modified organisms (GMOs), artificial colors, artificial sweeteners, or artificial preservatives.
CAUTIONS: Take under ongoing supervision with regular monitoring of blood chemistry, especially liver function. Do not take if you are pregnant or lactating.
All XYMOGEN® Formulas Meet or Exceed cGMP Quality Standards.
*These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease